Traditional Chinese Medicine for Epilepsy (Review) Li Q Chen X He L Zhou D
Introduction
Epilepsy is a common and chronic neurological disease. The etiologies of epilepsy are divers as structural, genetic, infectious, metabolic, immune, and unknown, which are proposed from International League Against Epilepsy classification arrangement in 2017 (Scheffer et al., 2017). The incidence and prevalence of epilepsy are higher in depression- and middle-income countries than in high-income countries, with approximately 80% of patients with epilepsy living in low- and center-income countries (Meyer et al., 2010; Beghi, 2020). The disease burden could be reduced past improving access to effective treatment (Beghi, 2020).
The pathogenesis of epilepsy is that abnormal electrical discharges derived from the brain including hippocampal, neocortical, cortico-thalamic and basal ganglia networks (Moshé et al., 2015). Though the causes of epilepsy are not totally clear, some possible mechanisms of epilepsy are proposed in many studies. Neurotransmitters, synapses, receptors, ion channels, inflammatory cytokines, immune systems, glial cells, oxidative stress, apoptosis, mitochondrial dysfunction, gene mutations, glycogen and glucocorticoids metabolisms are involved in the pathogenesis of epilepsy (He et al., 2021). Gamma amino butyric acid (GABA) is inhibitory neurotransmitter, and glutamate is excitatory one. Among iii types of GABA receptors, GABAA receptors command chloride ion influx, and GABAB receptors increase potassium outflow currents and reduce calcium entry. The activation of GABA receptors makes the inhibitory effect on neuronal membrane potential. Glutamate acts on alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionate (AMPA) receptors, kainite receptors, and Due north-methyl-D-aspartate (NMDA) receptors. The increased activity of NMDA receptors makes Ca2+ influx. Seizures and neuronal damages may occur when the imbalance of inhibitory and excitatory neural activity. Nicotine acetyl cholinergic (nACh) receptors and five-Hydroxytryptamine (5-HT) receptor besides control neuronal excitability and involve in epilepsy (Iha et al., 2017; Zhao et al., 2018). SCN1A, SCN2A, SCN3A, and SCN8A genes which individually encode voltage-gated sodium channels, that is Na5one.i, Na51.2, Na5one.iii, and NaV1.6, are related to early onset epilepsies (Brunklaus et al., 2020). Other mutations in ion channels, such as KCNMA1, KCNQ2, KCNT1, KCNQ3, CACNA1A, CLCN2, and HCN1-iv, bear upon the transportation of potassium, calcium, chloride, and cyclic nucleotide (He et al., 2021). Inflammation is the cause and the consequence of seizure, becoming a fell circle and leading epilepsy to develop and deteriorate (Vezzani et al., 2011). Both infectious and not-infectious inflammatory responses shared common allowed pathways and so contribute to epilepsy (Vezzani et al., 2016). Oxidative stress and mitochondrial dysfunction could besides be the causes and the results of genetic and acquired epilepsies by dissentious proteins, lipids, DNA, enzymes, and changing the neuronal excitability (Pearson-Smith and Patel, 2017). Oxidative stress and mitochondrial dysfunction induce apoptosis then led to neuronal death (Méndez-Armenta et al., 2014).
Epilepsy therapies contain anti-epilepsy medication, resective surgery and functional surgery, and medication is the major therapy. Currently canonical anti-epilepsy drugs mainly target voltage-gated ion channels such as sodium, potassium, and calcium channels, to modulate the electric firing of the neuron. For examples of this kind of drugs are phenytoin, carbamazepine, valproate, retigabine, ethosuximide, zonisamide, and and then on. Some drugs such as benzodiazepines, barbiturates, and tiagabine human action on GABA transporters and GABA receptors to raise the synaptic inhibition. Vigabatrin inhibit the GABA transaminase to reduce the metabolism of GABA. Some drugs human action on ionotropic glutamate receptors, such equally perampanel and topiramate human action on AMPA glutamate receptors or kainate receptors, and felbamate inhibit NMDA receptors, to suppress the synaptic excitation. Levetiracetam and brivaracetam bind to synaptic vesicle glycoprotein 2A (SV2A) to inhibit the release of glutamate (Wang and Chen, 2019).
Numerous herbal medicines, such as Ginkgo biloba and Huperzia serrata, take been reported to have antiepileptic or proconvulsant effects (Saxena and Nadkarni, 2011; Sahranavard et al., 2014; Ekstein, 2015; Kakooza-Mwesige, 2015; Shaikh, 2015; Xiao et al., 2015; Cai, 2017; Wei et al., 2017; Manchishi, 2018). The first anti-epilepsy medication resourced from institute is cannabidiol, which is approved by the United States Food and Drug Administration in 2018 for treating Dravet syndrome and Lennox-Gastaut syndrome (Samanta, 2019). Cannabidiol is a non-psychoactive agent of cannabis that is widely studied and proved for its efficacy and safety. Whether the mechanisms of its antiepileptic effect are not fully known, large amounts of clinical trials revealed its potential of medical utilize (Silvestro et al., 2019). But this new anti-epilepsy drug is expensive and less accessible in about countries because cannabis legalization and medical cannabis are nonetheless controversial issues.
Anti-epilepsy drugs take some agin furnishings on patients' quality of life. A latest review commodity generalized four challenges of anti-epilepsy drugs, including full general side furnishings, psychological challenges, social challenges, and economic challenges (Mutanana et al., 2020). The adverse furnishings of antiepileptic drugs include severe psychiatric, cognitive, behavioral, endocrine, and dermatological diseases and dysfunctions (Ekstein, 2015; Cai, 2017; Chen B. et al., 2017). The medications may affect the performance of patients' schoolwork, job, work, and may impede their marriage, and interpersonal human relationship. Low and suicidal ideation are related to increase the dose of anti-epilepsy drugs (Wen et al., 2010). For people who needs long-term handling of epilepsy, some of them surrender the unaffordable and inaccessible anti-epilepsy drugs. Those challenges brand patients escape from the handling with Western medicine, especially seen in the developing countries. Otherwise, even that at that place are many novel anti-epilepsy drugs adult in recent 20 years, virtually ane-tertiary of patients are defective appropriate seizure control due to pharmacoresistance (Wang and Chen, 2019). Currently, preventing epileptogenesis and treating comorbidities of epilepsy other than purely symptomatic command of seizures are the remaining challenges (Kobow et al., 2012; Terrone et al., 2016).
Natural medicine has found less side-effects and skillful efficacy in treating epilepsy. The mechanisms of natural medicine take been reported, including the regulation of synapses, receptors, and ion channels, the inhibition of inflammation, and the regulation of immune system. Natural medicine besides can correct the glial cells, meliorate mitochondrial dysfunction and oxidative stress, and regulate apoptosis (He et al., 2021). Chinese herbal medicine (CHM) has become a popular complementary and alternative medicine. The trend of seeking traditional Chinese medicine for treatment is caused by patients' fear of the side effects of surgery or Western medication (Ekstein, 2015; Kakooza-Mwesige, 2015). Traditional herbal medicine is also cheaper than mainstream therapy and could be more accessible to patients.
Chinese herbal medicine has been used to care for seizures and epilepsy for thousands of years. Traditional Chinese medicine is based on the theory that medicine and food come from the same sources. Therefore, people can consume herbal medicine in their daily diet. This practice is known as medical diet therapy. Medical diet therapy is the concept of combining nutrition and medicine to treat disease through eating (Wu and Liang, 2018).
The effectiveness of CHMs has also been demonstrated in recent studies. CHM is personalized medicine prescribed based on the constitution theory of Chinese medicine to maintain health and treat diseases (Li et al., 2019). Therefore, individuals may receive different herbal therapies for the same diagnosis.
The aim of this review is to summarize the clinical use and mechanisms of antiepileptic CHM and provide evidence for the efficacy of medical nutrition therapy, which warrants further exploration.
Materials and Methods
Mutual clinical used CHMs for treating epilepsy and seizure were searched and reviewed in PubMed and Cochrane Library. The various combinations of keywords included the terms "epilepsy," "seizure," "antiepileptic," "anticonvulsive," "Chinese herbal medicine," "Chinese herb," and each of the Latin names, English names, and scientific names of herbs. The search process is presented in Effigy 1. The sources of these antiepileptic herbs are summarized in Table 1 based on Taiwan'south official herbal pharmacopeia, third edition (Taiwan Herbal Pharmacopeia 3rd Edition Committee, 2019).
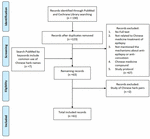
Figure 1. Flow diagram of the literature search procedure.
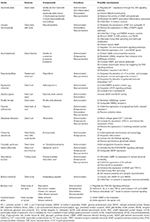
Table 1. Related functions and possible mechanisms of antiepileptic herbs.
Results and Discussion
Plants
Gastrodia elata
Gastrodia elata is a widely used traditional Chinese medicine for treating neurological disorders, such as headache, insomnia, and epilepsy (Zhan et al., 2016; Liu et al., 2018). G. elata has anticonvulsive, anti-inflammatory, neuroprotective, antiapoptosis, and antioxidative effects (Hsieh et al., 2001; Zhan et al., 2016; Liu et al., 2018). In a rat model of ferric-chloride-induced epileptic seizure, Vanillyl booze, a component of G. elata, suppressed seizures and lipid peroxidation. The pretreatment with either 200 mg/kg or 100 mg/kg Vanillyl alcohol significantly reduced the number of moisture dog shakes. The Vanillyl alcohol 200 mg/kg group had significantly greater suppress result on lipid peroxidation than the Vanillyl alcohol 100 mg/kg grouping and phenytoin 10 mg/kg group (Hsieh et al., 2000). In a rat model of kainic acid-induced epilepsy, Thousand. elata can suppress epileptic attacks past regulating the c-Jun Northward-terminal kinases (JNK) signal pathway and activator protein 1 (AP-1) expression. Both pre-treatment and post-treatment with G. elata modulated phosphorylated JNK and c-Jun protein. However, comparison pre-handling and mail service-treatment with G. elata, only pretreatment with G. elata inverse the levels of c-Fos protein, JNK protein, phosphorylated extracellular betoken-regulated kinase, and p38 proteins (Hsieh et al., 2007).
1 component of Thou. elata, gastrodin, did non act on ionotropic glutamate receptors to inhibit N-methyl-D-aspartate (NMDA) receptor–facilitated seizures but did reach neuroprotective effects through preventing NMDA excitotoxicity that is evaluated on rat hippocampal slice (Wong et al., 2016). Liu et al. reviewed the effects of Gastrodin, and summarized the mechanisms of Gastrodin including the modulation of neurotransmitters, antioxidative, anti-inflammatory, inhibition of microglial activation, regulating mitochondrial function, and up-regulating neurotrophins. Gastrodin has the ability to remainder the activity of gamma-aminobutyric acrid and glutamate (Liu et al., 2018). Gastrodin besides modulated mitogen-activated poly peptide kinase (MAPK)-associated inflammatory responses and inhibited Nav1.half dozen sodium currents, thereby reducing the severity of seizures that is proved by pentylenetetrazole (PTZ)-induced seizures mice model (Chen Fifty. et al., 2017; Shao et al., 2017). A study investigated and compared the pharmacokinetics of gratuitous gastrodin, parishin, and 1000. elata extract in rats. Parishin and Chiliad. elata extract had prolonged ti/2 compared with complimentary gastrodin in rat plasma, that is 3.09 ± 0.05 h, 7.52 ± 1.28 h and 1.13 ± 0.06 h respectively, indicating that Parishin and Chiliad. elata extract have longer action durations than free gastrodin does (Tang et al., 2015). Matias et al. (2016) reviewed diverse constituents of Grand. elata related to anticonvulsant activeness, including Grand. elata rhizome extracts, gastrodin, iv-Hydroxybenzyl alcohol, 4-Hydroxybenzaldehyde and analogs, vanillin, and vanillyl alcohol.
Research in 2020 revealed herb–drug interactions betwixt G. elata and carbamazepine (CBZ). G. elata reduced the autoinduction of CBZ and increased the plasma CBZ concentration (Yip et al., 2020). These studies revealed the values of M. elata as an anticonvulsant drug or adjuvant therapy. However, physicians should carefully consider drug dosage and side furnishings, such as itching rash and poor appetite, caused past the herb–drug interaction (Yip et al., 2020).
Uncaria rhynchophylla
Uncaria rhynchophylla (UR) and G. elata are commonly used in combination to treat convulsive disorder (Hsieh et al., 1999). They are considered an herb pair. In a kainic acid-treated rat model, UR has anticonvulsive and gratis radical scavenging activities and may have a synergistic effect when combined with 1000. elata that delays the onset of wet dog shakes, that is 63 min compared with 27 min in the command grouping, while 40 min in the Grand. elata group (Hsieh et al., 1999). Rhynchophylline is a component of UR that can treat the underexpression of macrophage migration inhibitory gene (MIF) and cyclophilin A in the frontal cortex and hippocampus in kainic acid-induced epilepsy rats. It showed UR group increased 3.1-fold MIF and ii.08-fold cyclophilin A while rhynchophylline group increased 2.75-fold MIF and 1.83-fold cyclophilin A in the frontal cortex; UR group increased ane.57-fold MIF and ane.35-fold cyclophilin A while rhynchophylline group increased 1.69-fold MIF and ane.26-fold cyclophilin A in the hippocampus, which were compared to the control group (Lo et al., 2010). Studies had reported that rhynchophylline can reduce epileptic seizures, a kainic acid-induced seizure rat model showed rhynchophylline can initiate c-Jun aminoterminal kinase phosphorylation (JNKp) in the MAPK signaling pathways (Hsu et al., 2013) as well as in a pilocarpine-induced status epilepticus rat model of temporal lobe epilepsy showed it can inhibit Navane.half dozen persistent sodium currents (INaP) and NMDA receptor currents (Shao et al., 2016). In kainic acrid-induced epileptic seizures rats, UR has neuroprotective furnishings through reducing glial fibrillary acidic protein and S100B protein expression and inhibiting receptors for avant-garde glycation cease products, non including GABAA and transient receptor potential vanilloid subtype 1 (TRPV1) receptors. UR has also been demonstrated to attenuate mossy fiber sprouting and astrocyte proliferation and forestall hippocampal neuron death, especially in the CA1 and CA3 areas (Lin and Hsieh, 2011; Liu et al., 2012; Tang et al., 2017). Furthermore, UR regulates toll-similar receptor and neurotrophin signaling pathways and inhibits the expressions of interleukin (IL)-1β and encephalon-derived neurotrophic factor (BDNF) genes in kainic acid-induced seizure rats' cortex and hippocampus (Ho et al., 2014).
Acori tatarinowii
Acori tatarinowii is a blazon of aquatic establish that is commonly used to treat neurological, cardiovascular, respiratory, and gastrointestinal diseases. A. tatarinowii decoction and its volatile oil accept been demonstrated that reduce seizure attacks in maximal electroshock (MES) model. The decoction of A. tatarinowii decreased convulsive rates in PTZ-induced seizure rats from 100% (normal saline command group) to 67% (dose 10 g/kg of decoction) while 33% in sodium valproate grouping. Volatile oil of A. tatarinowii could not decrease convulsive rates merely could reduce mortality rates of pentylenetetrazol-induced seizure rats from 92% (normal saline control group) to 40% (managed with dose 1.25 g/kg of volatile oil) (Liao et al., 2005). A major ingredient of A. tatarinowii, α-asarone, modulates GABAA receptors, enhances tonic GABAergic inhibition, and suppresses the excitability of CA1 hippocampal pyramidal neurons in PTZ and kainate mouse models (Huang et al., 2013). α-asarone and β-asarone increase the expression of neurotrophic factors, including nerve growth gene (NGF), BDNF, and glial-derived neurotrophic cistron (GDNF), in cultured rat astrocytes. The expression is partially activated by triggering the military camp-dependent protein kinase (PKA) signaling pathway (Lam et al., 2019). In the MES test and PTZ-induced seizures in mice models, eudesmin extracted from A. tatarinowii can increase GABA while reducing glutamate levels. Furthermore, eudesmin upregulates the expression of GABAA and glutamate decarboxylase 65 (GAD65) and modulates Caspase-iii and Bcl-2, both of which are related to neuron apoptosis (Liu et al., 2015).
Paeonia lactiflora
Paeonia lactiflora can suppress the height of c-Fos protein and increase transthyretin and phosphoglycerate mutase 1 expression in cobalt-treated mouse cerebrum, thereby exerting a neuroprotective issue on cerebral neurons (Kajiwara et al., 2008). Paeoniflorin, the major agile component of P. lactiflora. In a hyperthermia-induced seizure of immature rats' model, paeniflorin suppresses the top of glutamate-induced intracellular Catwo+, which is related to metabotropic glutamate receptor 5 (mGluR5) activation. The anticonvulsive effect of paeoniflorin is not associated with the release of GABA, the regulation of a-amino-3-hydroxy-5-methyl-iv-isoxazolpropionic acrid (AMPA), or the regulation of NMDA receptors. Information technology would be a possible herbal medicine for treating delirious seizure in children (Hino et al., 2012). Shosaiko-to-go-keishika-shyakuyaku-to is the Japanese Kampo medicine, and only Paeoniae radix, the main component of the formula, had significant inhibition effect of PTZ-induced EEG power spectrum changes (Sugaya et al., 1988).
Bupleurum chinense
Bupleurum chinense has various functions, including hepatoprotective, antitumor, antioxidant, antidepressant, anti-inflammatory, and anticonvulsant effects (Jiang et al., 2020). Saikosaponin a isolated from B. chinense showed anticonvulsive and neuroprotective furnishings past inhibiting NMDA receptor electric current, INorthward ap, and the mammalian target of rapamycin (mTOR) signaling pathway and increasing Kv4.2-mediated A-type voltage-gated potassium currents (Kv4.2-mediated IA) that proved by rat models (Yu et al., 2012; Ye et al., 2016; Hong et al., 2018). Saikosaponin tin can reduce the severity and elapsing of seizures, and prolong the latency of seizure in PTZ-induced rats (Ye et al., 2016). Some Chinese medicine formulas containing B. chinense, such as "Saiko-Keishi-To (Chai-Hu-Gui-Zhi-Tang)" and a modified formula of "Chaihu-Longu-Muli-Tang," take been reported to have anticonvulsant and antioxidant effects (Sugaya et al., 1985, 1988; Wu et al., 2002). The maintenance of calcium distribution and calcium binding state were shown in highly PTZ-sensitive snail neurons incubated in Saiko-keishi-to, and it indicated that Saiko-keishi-to has an inhibitory result on calcium shift and bounden country change (Sugaya et al., 1985). An open improver study performed modified formula of Chaihu-Longu-Muli-Tang for 20 refractory epilepsy and 20 benign epilepsy patients four months, and the formula subtract seizure frequency in refractory epileptics from thirteen.four ± three.4 to 10.vii ± 2.5 per calendar month (p-value was 0.084) that may be attribute to antioxidant furnishings with reducing serum malondialdehyde and copper-zinc superoxide dismutase (p < 0.05) while there are no statistically pregnant changes in beneficial epilepsy patients, that is considering only refractory epilepsy grouping has pregnant variation of lipid peroxidation compared to age-matched healthy command group (Wu et al., 2002).
Ziziphus jujuba
Ziziphus jujuba is ordinarily used to treat insomnia in traditional Chinese medicine. A report designed with the MES model and the PTZ model of rats indicated that Z. jujuba achieves anticonvulsant effects by increasing acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activity and the latency of myoclonic jerks, thereby preventing seizure attacks (Pahuja et al., 2011). The additional usage of hydroalcoholic excerpt of Z. jujuba tin heighten the anticonvulsant effects of phenytoin and phenobarbitone but not carbamazepine that is evaluated in MES-induced seizure rats (Pahuja et al., 2012).
Pinellia ternata
Pinellia ternata is generally used to care for ailments of the respiratory and gastrointestinal systems. A component of P. ternate, pinellia full alkaloids, is involved in the modulation of GABAergic systems through its increment of GABA and GAD65 expression, reduction of GABA transporter-i (GAT-1) and GABA transaminase (GABA-T) expression, and upregulation of GABAA receptor α5, δ, α4, and γ2 subunits in the hippocampal formation. Research in 2020 indicated that pinellia total alkaloids (PTA) may exert antiepileptogenic effects that reduce the occurrence of spontaneous recurrent seizures in pilocarpine-induced epileptic rats, and PTA 800 mg/kg grouping has the lowest frequencies of spontaneous recurrent seizures compared to PTA 400 mg/kg group and Topiramate lx mg/kg group (Deng et al., 2020).
Paeonia suffruticosa
Paeonol is extracted from the root bark of peony copse and is ordinarily used to activate blood circulation. A study in 2019 designed with 5 groups of PTZ-induced seizure rats, which are normal control grouping, epilepsy grouping, low-dose paeonol-treated group, medium-dose paeonol-treated group and high-dose paeonol-treated grouping that first explored the anticonvulsant effect of paeonol (Liu et al., 2019). Paeonol was determined to reduce the severity and elapsing of seizures and increase the latency of seizure. Furthermore, information technology protects hippocampal neurons from damage by reducing oxidative stress and inhibiting apoptosis in the CA1 areas while inhibiting the expression of the proapoptotic factor cleaved caspase-3. The seizure intensity was scored equally phase 0, no response; stage 1, facial movements and ear and whisker twitching; phase two, myoclonic convulsions without rearing; phase 3, myoclonic convulsions with rearing; stage four, tonic-clonic convulsions; stage v, generalized tonic-clonic seizures with loss of postural command; and phase 6, death. High-dose paeonol-treated group (60 mg/kg) reduce the seizure stage to 2.17 ± 0.41 compared to PTZ-kindled epilepsy group 4.67 ± 0.52 (Liu et al., 2019).
Stephania tetrandra
Tetrandrine is a voltage-gated Ca2+ aqueduct blocker isolated from S. tetrandra. A written report reported that tetrandrine regulates apoptosis and protects brain cells past increasing the expression of Bcl-two and reducing the expression of Bax. And tetrandrine could lessen the withdrawal symptoms such as weight loss induced by phenobarbital-dependency that proved by phenobarbital-withdrawn rat model (Han et al., 2015). Other studies in multidrug resistance cells and PTZ-induced seizure rats model revealed that tetrandrine tin reduce the antiepileptic drug resistance of phenytoin and valproate by reducing the expression of multidrug-resistant protein P-glycoprotein (P-gp) at the mRNA and poly peptide levels in the cortex and hippocampus, enhancing the efficacy of antiepileptic drugs. The seizure severity assessed by standards of Racine as grade IV and V were decreased in refractory epilepsy rats which were treated with tetrandrine (Chen et al., 2015).
Cistanche deserticola
Cistanche deserticola is a type of desert plants that grows in Cathay. Echinacoside is a chemical compound of C. deserticola. Pretreated x or 50 mg/kg echinacoside for 30 min on kainic acrid-induced seizures rats can enhance their neuronal survival and prevent epilepsy by inhibiting glutamate excitotoxicity and autophagy, suppressing inflammation, and activating poly peptide kinase B (Akt)/glycogen synthase kinase (GSK) 3β signaling. Therefore, information technology significantly increased seizure latency more than than ane h and decreased seizure severity (Lu et al., 2018a). A four-aminopyridine (4-AP)-induced epileptiform activeness with an in vitro rat hippocampal neurons' model report reported that echinacoside reduced spontaneous glutamate release, the frequency but not the amplitude of spontaneous excitatory postsynaptic currents, and the sustained repetitive firing of action potentials in hippocampal CA3 pyramidal neurons (Lu et al., 2018b).
Corydalis yanhusuo
Corydalis yanhusuo has analgesic effects. Furthermore, allaying, hypnotic, antihypertensive, antiepileptogenic, and anticonvulsant effects of C. yanhusuo have also been identified. Two studies used amygdala kindling seizures rats' model reported that DL-tetrahydropalmatine is a component that can inhibit amygdaloid dopamine release, thus reducing epilepsy attacks (Chang and Lin, 2001; Lin et al., 2002).
Salvia miltiorrhiza
Salvia miltiorrhiza is commonly used to ameliorate vascular circulation and has various functions, including anti-inflammatory, antioxidative, antiatherogenetic, antithrombotic, antihypertensive, antihyperlipidemic, antifibrotic, and antitumorous activities (Meim et al., 2019). Salvianolic acid B is a major water-soluble substance of S. miltiorrhiza. A study revealed that salvianolic acid B can reduce apoptosis and activate Akt/cAMP response element binding protein (CREB)/BDNF signaling pathways that contribute to neuronal survival and growth in the cortex and hippocampus, thus suppressing epilepsy in PTZ-kindled rats' model. PTZ-kindled rats treated with salvianolic acid B showed lower seizure stage 3.900 ± 0.718 compared to non-treated PTZ grouping 4.938 ± 0.250. Salvianolic acid B significantly decreased the full seizure times across the unabridged experiment from xix.150 ± v.851 to ix.600 ± three.515, and also significantly decrease the duration of seizures from 48.200 ± 5.782s to 29.950 ± iv.442s (Yu et al., 2019). Other studies take revealed that combining compound Danshen dripping pills (CDDP) with carbamazepine (CBZ) to treat kainic acid-induced temporal lobe epilepsy reduced seizure severity and frequency through antiapoptotic effects and the upregulation of GDNF expression in the CA3 area of the hippocampus. Compared to only CPPD or CBZ treatment group, the combination of CDDP with CBZ exerted a positive interaction effect that significantly decreased seizure phase, decreased the frequency of spontaneous recurrent seizures, and preserved the about number of surviving neurons (Jia et al., 2018).
Mucus
Ganoderma lucidum
In folklore, Ganoderma is considered a mysterious, magical, and precious Chinese medicine. A review in 2019 reported that the about commonly used Ganoderma are Grand. lucidum, M. applanatum, G. sinense, G. tsugae, G. capense, and G. boinense (Zhao et al., 2019). Nonetheless, G. lucidum is the traditional and nigh widely known species of Ganoderma.
A study harvested and cultured primary hippocampal neurons from rats, then established the epileptiform discharge hippocampal neuron model. The study indicated that One thousand. lucidum polysaccharides tin inhibit the accumulation of Ca2+ in hippocampal neurons and stimulate Ca2+/calmodulin-dependent poly peptide kinase II α (CaMK Two α) expression, thus reducing neuronal excitability (Wang et al., 2014). In an epileptiform discharge hippocampal neuron model, 1000. lucidum spores inhibit the expression of N-cadherin, which is related to mossy fiber sprouting and synaptic reconstruction, thus suppressing the neural excursion formed past mossy fiber sprouting. N-cadherin also promotes neurotrophin (NT)-iv expression, which is associated with neuron survival, inhibition of apoptosis, and synaptic plasticity, and thus protects hippocampal neurons (Wang et al., 2013). Ganoderic acid is the primary component of K. lucidum spores. In another epileptiform discharge hippocampal neuron model, Ganoderic acid prevents the apoptosis of hippocampal neurons and enhances the expression of BDNF and transient receptor potential approved 3 (TRPC3), which is involved in neuron plasticity and synaptic reconstruction, inhibits mossy fiber sprouting, and aids in the recovery of damaged neurons (Yang et al., 2016).
A retrospective written report in 2018 included 18 patients with epilepsy who were treated with G. lucidum spore powder therapy three times per day for 8 weeks. The study revealed that the powder reduced the weekly seizure frequency and the severity of each seizure episode (Wang et al., 2018). Further studies are required to confirm its efficacy in treating human being epilepsy.
Animals
Buthus martensii
Although scorpions take various levels of toxicity, they are a staple of traditional Asian street nutrient and medicinal wines from ancient times. Scorpions are normally used to treat neurological and musculoskeletal diseases, such as stroke, headache, seizure, and joint pain. B. martensii is the most abundant species of Asian scorpion and has been widely used in Chinese medicine since the Vocal dynasty of China. Antiepilepsy peptides (AEPs) are bioactive polypeptides extracted from its venom. AEP tin can easily cross the blood–brain barrier because of its low molecular weight (8.three kDa), and information technology exhibits anticonvulsant effects by binding with synaptosomal-associated protein (SNAP)-25 and NMDA (Wang et al., 2009). A study demonstrated that AEP can control neuronal excitability by selectively modifying voltage-gated sodium channels in master cortical neurons cultured from mice. AEP specially inhibits Navone.6 currents in homo embryonic kidney (HEK)-293 Cells, thus suppressing action potentials in neurons (Zhang et al., 2019).
Bombyx mori
Silkworms and their chrysalis are edible and high in protein. Infecting B. mori silkworms with the fungus Beauveria bassiana kills and dries the body of the silkworms. These infected silkworms are used as traditional Chinese medicine with reported anticonvulsant, anticoagulant, antitumor, antioxidant, antibacterial, antifungal, antiviral, hypoglycemic, and immunomodulatory furnishings (Hu et al., 2017, 2019). The anticonvulsant, hypnotic, and neurotrophic effects of some small molecule compounds, such as beauvericin and ammonium oxalate, take been explored (Hu et al., 2017). Several studies involving animal models have investigated the macromolecular compounds of B. mori, which had not been previously investigated. The poly peptide-rich extracts from B. mori were determined to human action mainly on the hippocampus CA1 region, and decreased seizure rates in MES-induced seizure mice and increased seizure and death latency in PTZ-induced seizure mice (Hu et al., 2019). The extracts protect neurons from oxidative damage and cell apoptosis by regulating the phosphoinositide 3-kinase (PI3K)/Akt signaling pathways in H2O2-stimulated PC12 cells (rat pheochromocytoma cells) in vitro (Hu et al., 2019). The extracts too achieve neuroprotective effects through reducing IL-1β, IL-4, and tumor necrosis gene (TNF)-α, increasing 5-HT and GABA, and reducing intracellular Ca2+ levels, preventing neuronal signaling, that was investigated on NGF-induced PC12 cells injured past glutamate (He et al., 2020).
Cryptotympana atrata
Cryptotympana atrata, cicada exuviae, is a usually used traditional Chinese herb in dermatological, ophthalmological, otorhinolaryngological, and neurological diseases. C. atrata can be cooked every bit porridge and soup, or made into tea for medical nutrition therapy. In a study of drug (PTZ, picrotoxin, or strychnine)-induced convulsions rat model, the extracts of C. atrata had anticonvulsive, allaying, and hypothermic effects; water extracts were more constructive than ethanol extracts (Hsieh et al., 1991).
Therefore, Chinese herbs (plant, fungi, and animals) exert anti-inflammatory, antioxidant, and neuroprotectant effects by acting on GABA, NMDA, and sodium channels, among others. The summarized possible mechanisms are presented in Tabular array 1. These effects are helpful for treating epileptic seizures. Yet, randomized, double-blind controlled clinical trials to ostend the antiepileptic effects and the efficacy in the treatment of epilepsy are lacking.
Evidence-Based Human Applications
To explore the evidence and reliability of Chinese medicine awarding on human, nosotros collect and review the human clinical trials. There are iv human clinical studies treating epilepsy with Chinese medicine that have been published. Three of the studies investigated the compounds of Chinese medicine, and i of the studies focused on a unique herb. Table ii describes the details of those studies.

Table ii. Characteristics of man clinical studies.
Saiko-ka-ryukotsu-borei-to (Chaihu-Longu-Muli-Tang) combined with Gastrodia elata and Uncaria rhynchophylla had antioxidant effects reducing the seizure frequency in refractory epilepsy patients from 13.4 ± 3.four to 10.vii ± 2.v every calendar month (Wu et al., 2002). A kind of anti-epilepsy sheathing, equanimous of Acorus tatarinowii, Arisaema cum Bile, Gastrodia elata, Pseudostellaria heterophylla, Poria cocos, Citrus reticulata, Pinellia ternata, Aquilaria sinensis, and Citrus aurantium, helped to control the electrical belch of the brain and improved the signs of epileptic electric belch shown by electroencephalography. Information technology effectively decreased the epilepsy frequency and the elapsing of attack for different types of epilepsy, including infantile spasm, autonomic, circuitous partial, holotonic-clonic, absence, localized Rolandic, psychomotor, myoclonus and indefinite types. The full constructive rate and recovery charge per unit of the intervention group are 83.33 and 54.3%, versus 51.88 and 38.4% in the control grouping, respectively (Ma et al., 2003). Dianxianning Pian is produced past the Chinese medicine manufacturing plant of China. The pill contains Valeriana jatamansi, Acorus tatarinowii, Uncaria rhynchophylla, Pharbitis nil, Euphorbia lathyris, Valeriana officinalis, and Nardostachys chinensis, and can command the frequency and severity of refractory epilepsy equally an adjunctive therapy. The average seizure rate decreased 37.84% in the intervention group while but xiii.18% in the control group, and the epilepsy frequency gradually reduced with increased treatment time (He et al., 2011).
A study in 2018 explored the efficacy of Ganoderma Lucidum spore pulverisation for treating epilepsy patients. The herb pulverization could reduce average weekly seizure frequency from 3.ane ± 0.eight to two.4 ± ane.2, but it didn't show meaning differences of duration of epilepsy and quality of life. The well-nigh common agin outcome is nausea, the second one is stomach discomfort, then the others are vomiting, dizziness, dry mouth, diarrhea, sore throat, and epistaxis in society (Wang et al., 2018).
Other Natural Medicine
A review article in 2018 reported eighteen anticonvulsant herbal agents, including Uncaria rhynchophylla, Gastrodia elata, Cannabis, Desmodium triflorum, Viscum album, Morus alba, Berberis integerrima, Mussaenda philippica, Justicia pectoralis, Gladiolus dalenii, Ficus religiosa, Withania somnifer, Lobelia nicotianaefolia, Marsilea quadrifolia, Passiflora incarnata, Mondia whitei, and Phytol (Manchishi, 2018). He et al. (2021) reviewed the bioactive compounds of natural drugs and categorized their antiepileptic mechanisms. There were 38 compounds could regulate neurotransmitters and synaptic function, 16 compounds regulate ion channels and the ion flow, 15 compounds modulate the immune system and exert anti-inflammatory effect, 5 compounds inhibit the activation of glial cells, 19 compounds exert anti-apoptotic and anti-oxidant result past protecting neurons from mitochondrial damage and oxidative stress, and 25 compounds care for epilepsy by other mechanisms (He et al., 2021). The review article mentioned natural drugs such every bit Salvia miltiorrhiza Bunge, Curcuma longa, Passiflora caerulea, Matricaria chamomilla, Scutellaria baicalensis, Bupleurum chinense, Plantago asiatica, Camellia sinensis, Withania somnifera, Maclura tinctoria, Radix astragali, Citrus reticulata Blanco, Cotinus coggygria, Gastrodia elata Blume, Radix bupleuri, Lantana camara, Curcuma longa, Rhododendron tomentosum, Matricaria chamomilla, Dennettia tripetala, Nigella sativa, Thymus vulgaris, Ginkgo biloba, Capsicum annuum, Crocus sativus, Uncaria rhynchophylla, Piper nigrum, Acorus tatarinowii, and so on. Among the natural medicine, cannabidiol inhibits neuronal excitability and exerts anticonvulsant backdrop, and it has been the first product fabricated directly from the cannabis constitute that approved by the Us Food and Drug Administration in 2018 (Lattanzi et al., 2018).
Herb–Drug Interaction
The combination therapy of anti-epileptic drugs and herbs are more and more popular and acceptable nowadays. 1 of the difficulties to ostend the herb–drug interaction is due to the circuitous ingredients of one herb, or in that location are many herbs in a formula of Chinese medicine. Some natural herbs interact with anti-epileptic drugs and then enhance the anti-convulsive furnishings were reported. At that place are few studies investigated the herb-drug interactions and the possible mechanisms (Pearl et al., 2011; He et al., 2021). In a PTZ-induced seizure mice model, Nobiletin and Clonazepam reduce seizure severity past regulating the remainder of glutamate and GABA, modulating GABAA and GAD 65, inhibiting apoptosis, inhibiting BDNF-TrkB signaling pathway and activating PI3K/Akt signaling pathway (Yang et al., 2018). The combination of Naringin and Phenytoin in PTZ-kindled rats could significantly subtract the seizure scores, elevate GABA and dopamine, subtract glutamate, against oxidation, and protect neurons in PTZ-induced seizure rats (Phani et al., 2018). Umbelliferone combined with phenobarbital or valproate elevate the threshold of electroconvulsions and heighten the anti-convulsive efficacy in MES-induced seizure mice model (Zagaja et al., 2015). In an oral CBZ rat model, sinapic acrid inhibits hepatic cytochrome P450 3A2, 2C11, and intestinal P-glycoprotein then increases the absorption of CBZ (Raish et al., 2019).
Those studies reported natural medicines, about of them are plants and herbs, can improve the efficacy of anti-epileptic drugs. In some studies, Chinese medicine showed the positive result in combined therapy with Western medicine that have been mentioned in this review article. There are rare studies explore the adverse effects of the combined therapy. It withal needs more well-designed studies to investigate the herb–drug interactions of Chinese medicine combined with anti-epileptic drugs considering of the insufficient evidences. Patients who use herbs as adjuvant therapy should inform their doctors to prevent the side-effects or complications that may cause past the potential herb–drug interactions.
Limitations
Nosotros reviewed Chinese medicine that is mainly used in clinical treatment in epilepsy, but other potential herbs may not be reviewed due to less references investigated. Some natural drugs include Chinese medicine are defective of large amounts of evidences to confirm the anti-epileptic furnishings. In addition, most studies are explored the efficacies and mechanisms of Chinese medicine in treating epilepsy but less mentioned its side effects. It nonetheless as well lack researchers to devote their efforts to the herb-drug interaction and side effects of Chinese medicine. As searching the databases, we found there are big portions of cell and animal models in studying the anti-epileptic effect of Chinese medicine, merely the human clinical trials are extremely deficient. For the safety and efficacy of Chinese medicine and natural drugs in testify-based practice, the further well-designed randomized controlled trials are promptly needed.
Conclusion
Anticonvulsive herbs used in clinical settings to treat epilepsy and seizure are discussed in the present article and their possible antiepileptic mechanisms, including anti-inflammation, antioxidation, GABAergic result enhancement, NMDA receptor and sodium aqueduct modulation, and neuroprotection, were described. Although some anticonvulsive effects of herbs take been reported, at that place are remaining the bug of the advisable handling time, dosage of herbs, and the long-term furnishings after the intervention. Larger sample size, high-quality randomized controlled clinical trials and adequate experimental evidences to ostend their antiepileptic furnishings are defective. Therefore, farther study is warranted.
Chinese medicine is holistic and can be personalized for individual patients based on their symptoms. Medical diet therapy using traditional Chinese medicine has spread globally. Herbal medicine is used equally adjunctive therapy or main therapy in some countries, particularly in Eastward. The time to come directions of the employ of herbal medicine are evidence-based do that needs further loftier-quality clinical trials for proving its efficacy and safety, and the concerns of herb-drug interaction equally combined therapy with Western medicine. Some Chinese herbs have been proved the anti-epilepsy furnishings and have the potential to improve the access to constructive treatment and to avert the adverse effects of Western medicine for epilepsy patients. The combined apply of different herbs exerts an anticonvulsant effect through various mechanisms. But for the safety of combined therapy with Western medicine, patients should consult and inform their physicians to notice the probable side effects acquired past herb–drug interactions.
Author Contributions
C-HL and C-LH: conceptualization. C-HL: data curation and writing—original typhoon grooming. C-LH: writing—review and editing. Both authors read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the "Chinese Medicine Research Middle, Mainland china Medical University" from The Featured Areas Research Center Plan within the framework of the Higher Education Sprout Project past the Ministry of Education in Taiwan (CMRC-Eye-0).
Conflict of Interest
The authors declare that the research was conducted in the absence of whatsoever commercial or financial relationships that could exist construed every bit a potential conflict of interest.
References
Brunklaus, A., Du, J., Steckler, F., Ghanty, I. I., Johannesen, K. M., and Fenger, C. D. (2020). Biological concepts in man sodium channel epilepsies and their relevance in clinical practice. Epilepsia 61, 387–399. doi: 10.1111/epi.16438
PubMed Abstract | CrossRef Full Text | Google Scholar
Cai, L. (2017). Traditional Chinese herbal medicine for epilepsy treatment should be administered according to the seizure blazon and epileptic syndrome. Health ix, 1211–1222. doi: 10.4236/health.2017.98087
CrossRef Full Text | Google Scholar
Chang, C. G., and Lin, One thousand. T. (2001). DL-Tetrahydropalmatine may act through inhibition of amygdaloid release of dopamine to inhibit an epileptic attack in rats. Neurosci. Lett. 307, 163–166. doi: x.1016/s0304-3940(01)01962-0
CrossRef Full Text | Google Scholar
Chen, B., Choi, H., Hirsch, 50. J., Katz, A., Legge, A., Buchsbaum, R., et al. (2017). Psychiatric and behavioral side furnishings of antiepileptic drugs in adults with epilepsy. Epilepsy Behav. 76, 24–31. doi: 10.1016/j.yebeh.2017.08.039
PubMed Abstract | CrossRef Full Text | Google Scholar
Chen, L., Liu, X., Wang, H., and Qu, Yard. (2017). Gastrodin attenuates pentylenetetrazole-induced seizures past modulating the mitogen-activated protein kinase-associated inflammatory responses in mice. Neurosci. Bull. 33, 264–272. doi: ten.1007/s12264-016-0084-z
PubMed Abstruse | CrossRef Full Text | Google Scholar
Chen, Y., Xiao, 10., Wang, C., Jiang, H., Hong, Z., and Xu, G. (2015). Benign effect of tetrandrine on refractory epilepsy via suppressing P-glycoprotein. Int. J. Neurosci. 125, 703–710. doi: ten.3109/00207454.2014.966821
PubMed Abstract | CrossRef Full Text | Google Scholar
Deng, C. X., Wu, Z. B., Chen, Y., and Yu, Z. M. (2020). Pinellia total alkaloids attune the GABAergic organisation in hippocampal germination on pilocarpine-induced epileptic rats. Chin. J. Integr. Med. 26, 138–145. doi: 10.1007/s11655-019-2944-7
PubMed Abstruse | CrossRef Total Text | Google Scholar
Han, B., Fu, P., Ye, Y., Zhang, H., and Wang, K. (2015). Protective effects of tetrandrine on encephalon cells in phenobarbital-dependent and -withdrawn rats. Mol. Med. Rep. xi, 1939–1944. doi: x.3892/mmr.2014.2997
PubMed Abstract | CrossRef Full Text | Google Scholar
He, Fifty., Wen, T., Yan, S., Li, R., Liu, Z., Ren, H., et al. (2011). Reevaluation of the effect of Dianxianning on seizure charge per unit of refractory epilepsy equally additive treatment in clinical practice. Forepart. Med. 5:229–234. doi: x.1007/s11684-011-0139-v
PubMed Abstruse | CrossRef Full Text | Google Scholar
He, L. Y., Hu, Thousand. B., Li, R. L., Zhao, R., Fan, 50. H., He, L., et al. (2021). Natural medicines for the treatment of epilepsy: bioactive components, pharmacology and machinery. Front. Pharmacol. 12:604040. doi: 10.3389/fphar.2021.604040
PubMed Abstract | CrossRef Full Text | Google Scholar
He, Fifty. Y., Hu, Thousand. B., Li, R. 50., Zhao, R., Fan, L. H., Wang, Fifty., et al. (2020). The effect of protein-rich excerpt from Bombyx batryticatus against glutamate-damaged PC12 cells via regulating gamma-aminobutyric acrid signaling pathway. Molecules 25:553. doi: 10.3390/molecules25030553
PubMed Abstract | CrossRef Full Text | Google Scholar
Hino, H., Takahashi, H., Suzuki, Y., Tanaka, J., Ishii, E., and Fukuda, M. (2012). Anticonvulsive effect of paeoniflorin on experimental febrile seizures in immature rats: possible application for febrile seizures in children. PLoS Ane 7:e42920. doi: 10.1371/journal.pone.0042920
PubMed Abstract | CrossRef Full Text | Google Scholar
Ho, T. Y., Tang, Northward. Y., Hsiang, C. Y., and Hsieh, C. 50. (2014). Uncaria rhynchophylla and rhynchophylline improved kainic acrid-induced epileptic seizures via IL-1beta and brain-derived neurotrophic factor. Phytomedicine 21, 893–900. doi: 10.1016/j.phymed.2014.01.011
PubMed Abstract | CrossRef Full Text | Google Scholar
Hong, Y., Deng, N., Jin, H. N., Xuan, Z. Z., Qian, Y. X., Wu, Z. Y., et al. (2018). Saikosaponin A modulates remodeling of Kv4.2-mediated A-type voltage-gated potassium currents in rat chronic temporal lobe epilepsy. Drug Des. Devel. Ther. 12, 2945–2958. doi: x.2147/DDDT.S166408
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hsieh, C. L., Chang, C. H., Chiang, S. Y., Li, T. C., Tang, N. Y., Pon, C. Z., et al. (2000). Anticonvulsive and complimentary radical scavenging activities of vanillyl booze in ferric chloride-induced epileptic seizures in Sprague-Dawley rats. Life Sci. 67, 1185–1195. doi: ten.1016/s0024-3205(00)00706-ii
CrossRef Full Text | Google Scholar
Hsieh, C. Fifty., Chiang, S. Y., Cheng, K. South., Lin, Y. H., Tang, Northward. Y., Lee, C. J., et al. (2001). Anticonvulsive and complimentary radical scavenging activities of Gastrodia elata Bl. in kainic acrid-treated rats. Am. J. Chin. Med. 29, 331–341. doi: 10.1142/S0192415X01000356
PubMed Abstruse | CrossRef Total Text | Google Scholar
Hsieh, C. 50., Lin, J. J., Chiang, S. Y., Su, S. Y., Tang, Due north. Y., Lin, G. G., et al. (2007). Gastrodia elata modulated activator protein one via c-Jun N-terminal kinase signaling pathway in kainic acid-induced epilepsy in rats. J. Ethnopharmacol. 109, 241–247. doi: 10.1016/j.jep.2006.07.024
PubMed Abstract | CrossRef Full Text | Google Scholar
Hsieh, C. 50., Tang, N. Y., Chiang, South. Y., Hsieh, C. T., and Lin, J. G. (1999). Anticonvulsive and gratis radical scavenging actions of 2 herbs, Uncaria rhynchophylla (MIQ) Jack and Gastrodia elata Bl., in kainic acid-treated rats. Life Sci. 65, 2071–2082. doi: 10.1016/s0024-3205(99)00473-7
CrossRef Total Text | Google Scholar
Hsieh, M. T., Peng, W. H., Yeh, F. T., Tsai, H. Y., and Chang, Y. Due south. (1991). Studies on the anticonvulsive, allaying and hypothermic effects of Periostracum Cicadae extracts. J. Ethnopharmacol. 35, 83–90. doi: 10.1016/0378-8741(91)90136-2
CrossRef Total Text | Google Scholar
Hsu, H. C., Tang, Due north. Y., Liu, C. H., and Hsieh, C. L. (2013). Antiepileptic effect of Uncaria rhynchophylla and rhynchophylline involved in the initiation of c-Jun N-last kinase phosphorylation of MAPK signal pathways in acute seizures of kainic acid-treated rats. Evid. Based Complement. Alternat. Med. 2013:961289. doi: 10.1155/2013/961289
PubMed Abstract | CrossRef Total Text | Google Scholar
Hu, Thou., Liu, Y., He, 50., Yuan, Ten., Peng, W., and Wu, C. (2019). Antiepileptic furnishings of poly peptide-rich extract from Bombyx batryticatus on mice and its protective effects against H2O2-induced oxidative damage in PC12 cells via regulating PI3K/Akt signaling pathways. Oxid. Med. Jail cell. Longev. 2019:7897584. doi: 10.1155/2019/7897584
PubMed Abstract | CrossRef Total Text | Google Scholar
Hu, M., Yu, Z., Wang, J., Fan, W., Liu, Y., Li, J., et al. (2017). Traditional uses, origins, chemical science and pharmacology of Bombyx batryticatus: a review. Molecules 22:1779. doi: 10.3390/molecules22101779
PubMed Abstruse | CrossRef Full Text | Google Scholar
Huang, C., Li, W. G., Zhang, X. B., Wang, L., Xu, T. 50., Wu, D., et al. (2013). Alpha-asarone from Acorus gramineus alleviates epilepsy by modulating A-type GABA receptors. Neuropharmacology 65, 1–11. doi: 10.1016/j.neuropharm.2012.09.001
PubMed Abstract | CrossRef Full Text | Google Scholar
Iha, H. A., Kunisawa, North., Shimizu, Southward., Tokudome, Thou., Mukai, T., Kinboshi, Chiliad., et al. (2017). Nicotine elicits convulsive seizures by activating amygdalar neurons. Front. Pharmacol. eight:57. doi: 10.3389/fphar.2017.00057
PubMed Abstract | CrossRef Full Text | Google Scholar
Jia, C., Han, Southward., Wei, L., Dang, 10., Niu, Q., Chen, G., et al. (2018). Protective upshot of compound Danshen (Salvia miltiorrhiza) dripping pills alone and in combination with carbamazepine on kainic acrid-induced temporal lobe epilepsy and cognitive impairment in rats. Pharm. Biol. 56, 217–224. doi: 10.1080/13880209.2018.1432665
PubMed Abstract | CrossRef Full Text | Google Scholar
Jiang, H., Yang, Fifty., Hou, A., Zhang, J., Wang, S., Human, W., et al. (2020). Phytology, traditional uses, phytochemistry, belittling methods, processing, pharmacology and pharmacokinetics of Bupleuri Radix: a systematic review. Biomed. Pharmacother. 131:110679. doi: ten.1016/j.biopha.2020.110679
PubMed Abstract | CrossRef Full Text | Google Scholar
Kajiwara, Grand., Sunaga, K., Tsuda, T., Sugaya, A., Sugaya, E., and Kimura, 1000. (2008). Peony root extract upregulates transthyretin and phosphoglycerate mutase in mouse cobalt focus seizure. Biochem. Biophys. Res. Commun. 371, 375–379. doi: 10.1016/j.bbrc.2008.04.094
PubMed Abstract | CrossRef Full Text | Google Scholar
Kakooza-Mwesige, A. (2015). The importance of botanical treatments in traditional societies and challenges in developing countries. Epilepsy Behav. 52, 297–307. doi: 10.1016/j.yebeh.2015.06.017
PubMed Abstruse | CrossRef Total Text | Google Scholar
Kobow, Thousand., Auvin, South., Jensen, F., Löscher, W., Mody, I., Potschka, H., et al. (2012). Finding a meliorate drug for epilepsy: antiepileptogenesis targets. Epilepsia 53, 1868–1876. doi: x.1111/j.1528-1167.2012.03716.ten
PubMed Abstract | CrossRef Full Text | Google Scholar
Lam, K. Y. C., Wu, Q. Y., Hu, W. H., Yao, P., Wang, H. Y., Dong, T. T. X., et al. (2019). Asarones from Acori Tatarinowii Rhizoma stimulate expression and secretion of neurotrophic factors in cultured astrocytes. Neurosci. Lett. 707:134308. doi: x.1016/j.neulet.2019.134308
PubMed Abstract | CrossRef Total Text | Google Scholar
Lattanzi, S., Brigo, F., Trinka, Eastward., Zaccara, M., Cagnetti, C., Del Giovane, C., et al. (2018). Efficacy and safety of cannabidiol in epilepsy: a systematic review and meta-assay. Drugs 78, 1791–1804. doi: x.1007/s40265-018-0992-5
PubMed Abstract | CrossRef Total Text | Google Scholar
Li, L., Yao, H., Wang, J., Li, Y., and Wang, Q. (2019). The role of Chinese medicine in health maintenance and affliction prevention: application of constitution theory. Am. J. Chin. Med. 47, 495–506. doi: ten.1142/S0192415X19500253
PubMed Abstract | CrossRef Full Text | Google Scholar
Liao, W. P., Chen, L., Yi, Y. H., Sun, Westward. W., Gao, K. K., Su, T., et al. (2005). Report of antiepileptic effect of extracts from Acorus tatarinowii Schott. Epilepsia 46, 21–24. doi: x.1111/j.0013-9580.2005.461007.x
PubMed Abstract | CrossRef Total Text | Google Scholar
Lin, M. T., Wang, J. J., and Young, M. Due south. (2002). The protective effect of dl-tetrahydropalmatine against the evolution of amygdala kindling seizures in rats. Neurosci. Lett. 320, 113–116. doi: x.1016/s0304-3940(01)02508-3
CrossRef Full Text | Google Scholar
Lin, Y. W., and Hsieh, C. L. (2011). Oral Uncaria rhynchophylla (UR) reduces kainic acid-induced epileptic seizures and neuronal decease accompanied past attenuating glial cell proliferation and S100B proteins in rats. J. Ethnopharmacol. 135, 313–320. doi: 10.1016/j.jep.2011.03.018
PubMed Abstract | CrossRef Full Text | Google Scholar
Liu, C. H., Lin, Y. W., Tang, N. Y., Liu, H. J., and Hsieh, C. L. (2012). Neuroprotective effect of Uncaria rhynchophylla in kainic acid-induced epileptic seizures by modulating hippocampal mossy fiber sprouting, neuron survival, astrocyte proliferation, and S100B expression. Evid. Based Complement. Alternat. Med. 2012:194790. doi: 10.1155/2012/194790
PubMed Abstruse | CrossRef Full Text | Google Scholar
Liu, D. H., Agbo, E., Zhang, Due south. H., and Zhu, J. Fifty. (2019). Anticonvulsant and neuroprotective effects of paeonol in epileptic rats. Neurochem Res. 44, 2556–2565. doi: 10.1007/s11064-019-02874-6
PubMed Abstruse | CrossRef Full Text | Google Scholar
Liu, H., Vocal, Z., Liao, D. K., Zhang, T. Y., Liu, F., Zhuang, 1000., et al. (2015). Anticonvulsant and sedative effects of eudesmin isolated from Acorus tatarinowii on mice and rats. Phytother. Res. 29, 996–1003. doi: 10.1002/ptr.5337
PubMed Abstruse | CrossRef Total Text | Google Scholar
Liu, Y., Gao, J., Peng, Chiliad., Meng, H., Ma, H., Cai, P., et al. (2018). A review on cardinal nervous arrangement effects of gastrodin. Front. Pharmacol. 9:24. doi: 10.3389/fphar.2018.00024
PubMed Abstract | CrossRef Total Text | Google Scholar
Lo, W. Y., Tsai, F. J., Liu, C. H., Tang, Northward. Y., Su, Southward. Y., Lin, S. Z., et al. (2010). Uncaria rhynchophylla upregulates the expression of MIF and cyclophilin A in kainic acid-induced epilepsy rats: a proteomic analysis. Am. J. Chin. Med. 38, 745–759. doi: x.1142/S0192415X10008214
PubMed Abstract | CrossRef Full Text | Google Scholar
Lu, C. Due west., Hsieh, H. L., Lin, T. Y., Hsieh, T. Y., Huang, S. Grand., and Wang, South. J. (2018a). Echinacoside, an active constituent of Cistanche Herba, exerts a neuroprotective effect in a kainic acid rat model by inhibiting inflammatory processes and activating the Akt/GSK3β pathway. Biol. Pharm. Bull. 41, 1685–1693. doi: 10.1248/bpb.b18-00407
PubMed Abstract | CrossRef Full Text | Google Scholar
Lu, C. W., Huang, Due south. Yard., Lin, T. Y., and Wang, S. J. (2018b). Echinacoside, an agile constituent of Herba Cistanche, suppresses epileptiform activity in hippocampal CA3 pyramidal neurons. Korean J. Physiol. Pharmacol. 22, 249–255. doi: 10.4196/kjpp.2018.22.3.249
PubMed Abstract | CrossRef Full Text | Google Scholar
Ma, R., Li, South., Li, X., Hu, S., Sunday, X., Liu, Y., et al. (2003). Clinical ascertainment on 930 child epilepsy cases treated with anti-epilepsy capsules. J. Tradit. Chin. Med. 23, 109–112.
Google Scholar
Matias, M., Silvestre, South., Falcao, A., and Alves, Thousand. (2016). Gastrodia elata and epilepsy: rationale and therapeutic potential. Phytomedicine 23, 1511–1526. doi: 10.1016/j.phymed.2016.09.001
PubMed Abstract | CrossRef Total Text | Google Scholar
Meim, X. D., Cao, Y. F., Che, Y. Y., Li, J., Shang, Z. P., Zhao, Westward. J., et al. (2019). Danshen: a phytochemical and pharmacological overview. Mentum. J. Nat. Med. 17, 59–80. doi: 10.1016/S1875-5364(19)30010-X
CrossRef Full Text | Google Scholar
Méndez-Armenta, Thousand., Nava-Ruíz, C., Juárez-Rebollar, D., Rodríguez-Martínez, East., and Gómez, P. Y. (2014). Oxidative stress associated with neuronal apoptosis in experimental models of epilepsy. Oxid. Med. Cell. Longev. 2014:293689. doi: 10.1155/2014/293689
PubMed Abstract | CrossRef Full Text | Google Scholar
Meyer, A. C., Dua, T., Ma, J., Saxena, Southward., and Birbeck, One thousand. (2010). Global disparities in the epilepsy handling gap: a systematic review. Bull. World Wellness Organ. 88, 260–266. doi: 10.2471/BLT.09.064147
PubMed Abstruse | CrossRef Total Text | Google Scholar
Mutanana, Northward., Tsvere, Yard., and Chiweshe, M. K. (2020). Full general side effects and challenges associated with anti-epilepsy medication: a review of related literature. Afr. J. Prim. Health Care Fam. Med. thirty, e1–e5. doi: x.4102/phcfm.v12i1.2162
PubMed Abstract | CrossRef Full Text | Google Scholar
Pahuja, M., Kleekal, T., Reeta, One thousand. H., Tripathi, Yard., and Gupta, Y. M. (2012). Interaction profile of Zizyphus jujuba with phenytoin, phenobarbitone, and carbamazepine in maximal electroshock-induced seizures in rats. Epilepsy Behav. 25, 368–373. doi: 10.1016/j.yebeh.2012.08.014
PubMed Abstract | CrossRef Full Text | Google Scholar
Pahuja, M., Mehla, J., Reeta, 1000. H., Joshi, S., and Gupta, Y. K. (2011). Hydroalcoholic extract of Zizyphus jujuba ameliorates seizures, oxidative stress, and cognitive impairment in experimental models of epilepsy in rats. Epilepsy Behav. 21, 356–363. doi: x.1016/j.yebeh.2011.05.013
PubMed Abstruse | CrossRef Full Text | Google Scholar
Pearl, P. L., Drillings, I. M., and Conry, J. A. (2011). Herbs in epilepsy: evidence for efficacy, toxicity, and interactions. Semin. Pediatr. Neurol. xviii, 203–208. doi: 10.1016/j.spen.2011.06.007
PubMed Abstract | CrossRef Full Text | Google Scholar
Phani, Thou. K., Annapurna, A., Lakshmi, S. Northward. R., Ravi, C. S. R. D., Abutalaha, Chiliad., and Srikanth, I. (2018). Naringin in a combined therapy with phenytoin on pentylenetetrazole-induced kindling in rats. Epilepsy Behav. 89, 159–168. doi: 10.1016/j.yebeh.2018.x.006
PubMed Abstract | CrossRef Full Text | Google Scholar
Raish, M., Ahmad, A., Ansari, M. A., Alkharfy, K. M., Ahad, A., Al-Jenoobi, F. I., et al. (2019). Effects of sinapic acid on hepatic cytochrome P450 3A2, 2C11, and intestinal P-glycoprotein on the pharmacokinetics of oral carbamazepine in rats: potential food/herb-drug interaction. Epilepsy Res. 153, 14–18. doi: 10.1016/j.eplepsyres.2019.03.012
PubMed Abstract | CrossRef Full Text | Google Scholar
Sahranavard, S., Ghafari, South., and Mosaddegh, M. (2014). Medicinal plants used in Iranian traditional medicine to treat epilepsy. Seizure 23, 328–332. doi: x.1016/j.seizure.2014.01.013
PubMed Abstract | CrossRef Full Text | Google Scholar
Scheffer, I. E., Berkovic, South., Capovilla, G., Connolly, One thousand. B., French, J., Guilhoto, 50., et al. (2017). ILAE classification of the epilepsies: position paper of the ILAE committee for nomenclature and terminology. Epilepsia 58, 512–521. doi: 10.1111/epi.13709
PubMed Abstruse | CrossRef Total Text | Google Scholar
Shaikh, Chiliad. F. (2015). A review on natural therapy for seizure disorders. Pharm. Pharmacol. Int. J. 3, 255–259. doi: 10.15406/ppij.2015.03.00051
CrossRef Full Text | Google Scholar
Shao, H., Yang, Y., Mi, Z., Zhu, G. X., Qi, A. P., Ji, Due west. G., et al. (2016). Anticonvulsant effect of rhynchophylline involved in the inhibition of persistent sodium electric current and NMDA receptor current in the pilocarpine rat model of temporal lobe epilepsy. Neuroscience 337, 355–369. doi: x.1016/j.neuroscience.2016.09.029
PubMed Abstract | CrossRef Full Text | Google Scholar
Shao, H., Yang, Y., Qi, A. P., Hong, P., Zhu, 1000. X., Cao, 10. Y., et al. (2017). Gastrodin reduces the severity of status epilepticus in the rat pilocarpine model of temporal lobe epilepsy by inhibiting Nav1.6 sodium currents. Neurochem. Res. 42, 360–374. doi: 10.1007/s11064-016-2079-6
PubMed Abstract | CrossRef Total Text | Google Scholar
Silvestro, South., Mammana, S., Cavalli, E., Bramanti, P., and Mazzon, Eastward. (2019). Use of cannabidiol in the treatment of epilepsy: efficacy and security in clinical trials. Molecules 12:1459. doi: 10.3390/molecules24081459
PubMed Abstract | CrossRef Full Text | Google Scholar
Sugaya, A., Tsuda, T., Yasuda, Yard., Sugaya, Eastward., and Onozuka, Grand. (1985). Issue of Chinese herbal medicine "saiko-keishi-to" on intracellular calcium and poly peptide behavior during pentylenetetrazole-induced bursting activeness in snail neurons. Planta Med. 51, 2–6. doi: x.1055/due south-2007-969377
PubMed Abstruse | CrossRef Full Text | Google Scholar
Sugaya, E., Ishige, A., Sekiguchi, M., Iizuka, S., Ito, K., Sugimoto, A., et al. (1988). Inhibitory effect of TJ-960 (SK) on pentylenetetrazol-induced EEG ability spectrum changes. Epilepsy Res. 2, 27–31. doi: x.1016/0920-1211(88)90006-x
CrossRef Full Text | Google Scholar
Taiwan Herbal Pharmacopeia third Edition Committee (2019). Taiwan Herbal Pharmacopeia, English Version, 3rd Edn. Taiwan: Ministry Health and Welfare, x–388.
Google Scholar
Tang, C., Wang, Fifty., Liu, X., Cheng, Grand., Qu, Y., and Xiao, H. (2015). Comparative pharmacokinetics of gastrodin in rats later intragastric assistants of free gastrodin, parishin and Gastrodia elata extract. J. Ethnopharmacol. 176, 49–54. doi: x.1016/j.jep.2015.x.007
PubMed Abstract | CrossRef Full Text | Google Scholar
Tang, Due north. Y., Lin, Y. W., Ho, T. Y., Cheng, C. Y., Chen, C. H., and Hsieh, C. L. (2017). Long-term intake of Uncaria rhynchophylla reduces S100B and RAGE protein levels in kainic acrid-induced epileptic seizures rats. Evid. Based Complement. Alternat. Med. 2017:9732854. doi: ten.1155/2017/9732854
PubMed Abstract | CrossRef Full Text | Google Scholar
Vezzani, A., Fujinami, R. S., White, H. S., Preux, P. M., Blümcke, I., Sander, J. W., et al. (2016). Infections, inflammation and epilepsy. Acta Neuropathol. 131, 211–234. doi: 10.1007/s00401-015-1481-5
PubMed Abstract | CrossRef Full Text | Google Scholar
Wang, Grand. H., Li, X., Cao, W. H., Li, J., and Wang, 50. H. (2018). A retrospective study of Ganoderma lucidum spore powder for patients with epilepsy. Medicine 97:e10941. doi: 10.1097/Medico.0000000000010941
PubMed Abstract | CrossRef Full Text | Google Scholar
Wang, Southward. Q., Li, 10. J., Qiu, H. B., Jiang, Z. Thousand., Simon, 1000., Ma, 10. R., et al. (2014). Anti-epileptic result of Ganoderma lucidum polysaccharides by inhibition of intracellular calcium accumulation and stimulation of expression of CaMKII blastoff in epileptic hippocampal neurons. PLoS One nine:e102161. doi: ten.1371/periodical.pone.0102161
PubMed Abstract | CrossRef Full Text | Google Scholar
Wang, S. Q., Li, X. J., Zhou, S., Sun, D. Ten., Wang, H., Cheng, P. F., et al. (2013). Intervention effects of Ganoderma lucidum spores on epileptiform belch hippocampal neurons and expression of neurotrophin-4 and N-cadherin. PLoS Ane eight:e61687. doi: 10.1371/journal.pone.0061687
PubMed Abstract | CrossRef Full Text | Google Scholar
Wang, Y., and Chen, Z. (2019). An update for epilepsy enquiry and antiepileptic drug evolution: toward precise circuit therapy. Pharmacol. Ther. 201, 77–93. doi: x.1016/j.pharmthera.2019.05.010
PubMed Abstruse | CrossRef Full Text | Google Scholar
Wang, Z., Wang, Westward., Shao, Z., Gao, B., Li, J., Ma, J., et al. (2009). Eukaryotic expression and purification of anti-epilepsy peptide of Buthus martensii Karsch and its poly peptide interactions. Mol. Cell. Biochem. 330, 97–104. doi: 10.1007/s11010-009-0104-7
PubMed Abstract | CrossRef Full Text | Google Scholar
Wei, L., Tongtong, Thousand., Zhenxiang, P., Yashu, L., Jiayin, L., and Li, B. (2017). The effects of herbal medicine on epilepsy. Oncotarget 8, 48385–48397. doi: 10.18632/oncotarget.16801
PubMed Abstruse | CrossRef Total Text | Google Scholar
Wen, Ten., Meador, Grand. J., Loring, D. W., Eisenschenk, South., Segal, R., and Hartzema, A. Yard. (2010). Is antiepileptic drug use related to depression and suicidal ideation among patients with epilepsy? Epilepsy Behav. 19, 494–500. doi: ten.1016/j.yebeh.2010.08.030
PubMed Abstract | CrossRef Full Text | Google Scholar
Wong, Southward. B., Hung, Due west. C., and Min, M. Y. (2016). The role of gastrodin on hippocampal neurons afterwards Northward-Methyl-D-Aspartate excitotoxicity and experimental temporal lobe seizures. Chin. J. Physiol. 59, 156–164. doi: ten.4077/CJP.2016.BAE385
PubMed Abstract | CrossRef Full Text | Google Scholar
Wu, H. M., Liu, C. S., Tsai, J. J., Ko, 50. Y., and Wei, Y. H. (2002). Antioxidant and anticonvulsant consequence of a modified formula of chaihu-longu-muli-tang. Am. J. Chin. Med. 30, 339–346. doi: 10.1142/S0192415X02000235
PubMed Abstract | CrossRef Total Text | Google Scholar
Wu, Q., and Liang, Ten. (2018). Food therapy and medical diet therapy of traditional Chinese medicine. Clin. Nutr. Exp. eighteen, one–v. doi: 10.1016/j.yclnex.2018.01.001
CrossRef Full Text | Google Scholar
Xiao, F., Yan, B., Chen, L., and Zhou, D. (2015). Review of the use of botanicals for epilepsy in complementary medical systems–Traditional Chinese Medicine. Epilepsy Behav. 52, 281–289. doi: 10.1016/j.yebeh.2015.04.050
PubMed Abstract | CrossRef Full Text | Google Scholar
Yang, Z. West., Wu, F., and Zhang, Southward. L. (2016). Effects of ganoderic acids on epileptiform discharge hippocampal neurons: insights from alterations of BDNF. TRPC3 and apoptosis. Pharmazie 71, 340–344. doi: 10.1691/ph.2016.5889
CrossRef Total Text | Google Scholar
Ye, M., Bi, Y. F., Ding, 50., Zhu, Due west. W., and Gao, Due west. (2016). Saikosaponin a functions as anti-epileptic event in pentylenetetrazol induced rats through inhibiting mTOR signaling pathway. Biomed. Pharmacother. 81, 281–287. doi: 10.1016/j.biopha.2016.04.012
PubMed Abstract | CrossRef Full Text | Google Scholar
Yip, 1000. L., Zhou, 10., Chook, P., Leung, P. C., Schachter, S., Mok, 5. C. T., et al. (2020). Herb-drug interaction of gastrodiae rhizoma on carbamazepine: a pharmacokinetic study in rats. Epilepsy Res. 165:106376. doi: ten.1016/j.eplepsyres.2020.106376
PubMed Abstract | CrossRef Full Text | Google Scholar
Yu, 10., Guan, Q., Wang, Y., Shen, H., Zhai, L., Lu, X., et al. (2019). Anticonvulsant and anti-apoptosis effects of salvianolic acid B on pentylenetetrazole-kindled rats via AKT/CREB/BDNF signaling. Epilepsy Res. 154, 90–96. doi: x.1016/j.eplepsyres.2019.05.007
PubMed Abstract | CrossRef Full Text | Google Scholar
Yu, Y. H., Xie, Due west., Bao, Y., Li, H. G., Hu, S. J., and Xing, J. L. (2012). Saikosaponin a mediates the anticonvulsant backdrop in the HNC models of AE and SE past inhibiting NMDA receptor current and persistent sodium current. PLoS One 7:e50694. doi: 10.1371/periodical.pone.0050694
PubMed Abstract | CrossRef Full Text | Google Scholar
Zagaja, Chiliad., Andres-Mach, One thousand., Skalicka-Woźniak, K., Rękas, A. R., Kondrat-Wróbel, M. Westward., Gleńsk, M., et al. (2015). Assessment of the combined handling with umbelliferone and iv classical antiepileptic drugs confronting maximal electroshock-induced seizures in mice. Pharmacology 96, 175–180. doi: 10.1159/000438704
PubMed Abstract | CrossRef Full Text | Google Scholar
Zhan, H. D., Zhou, H. Y., Sui, Y. P., Du, X. L., Wang, Due west. H., Dai, L., et al. (2016). The rhizome of Gastrodia elata Blume–an ethnopharmacological review. J. Ethnopharmacol. 189, 361–385. doi: ten.1016/j.jep.2016.06.057
PubMed Abstruse | CrossRef Full Text | Google Scholar
Zhang, F., Wu, Y., Zou, 10., Tang, Q., Zhao, F., and Cao, Z. (2019). BmK AEP, an anti-epileptic peptide distinctly affects the gating of brain subtypes of voltage-gated sodium channels. Int. J. Mol. Sci. 20:729. doi: 10.3390/ijms20030729
PubMed Abstruse | CrossRef Full Text | Google Scholar
Zhao, C., Zhang, C., Xing, Z., Ahmad, Z., Li, J. Southward., and Chang, Chiliad. W. (2019). Pharmacological effects of natural Ganoderma and its extracts on neurological diseases: a comprehensive review. Int. J. Biol. Macromol. 121, 1160–1178. doi: 10.1016/j.ijbiomac.2018.10.076
PubMed Abstruse | CrossRef Total Text | Google Scholar
Zhao, H., Lin, Y., Chen, S., Li, X., and Huo, H. (2018). 5-HT3 receptors: a potential therapeutic target for epilepsy. Curr. Neuropharmacol. 16, 29–36. doi: 10.2174/1570159X15666170508170412
PubMed Abstruse | CrossRef Full Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fnins.2021.682821/full
0 Response to "Traditional Chinese Medicine for Epilepsy (Review) Li Q Chen X He L Zhou D"
Postar um comentário